Cell-matrix Interactions
Project Description
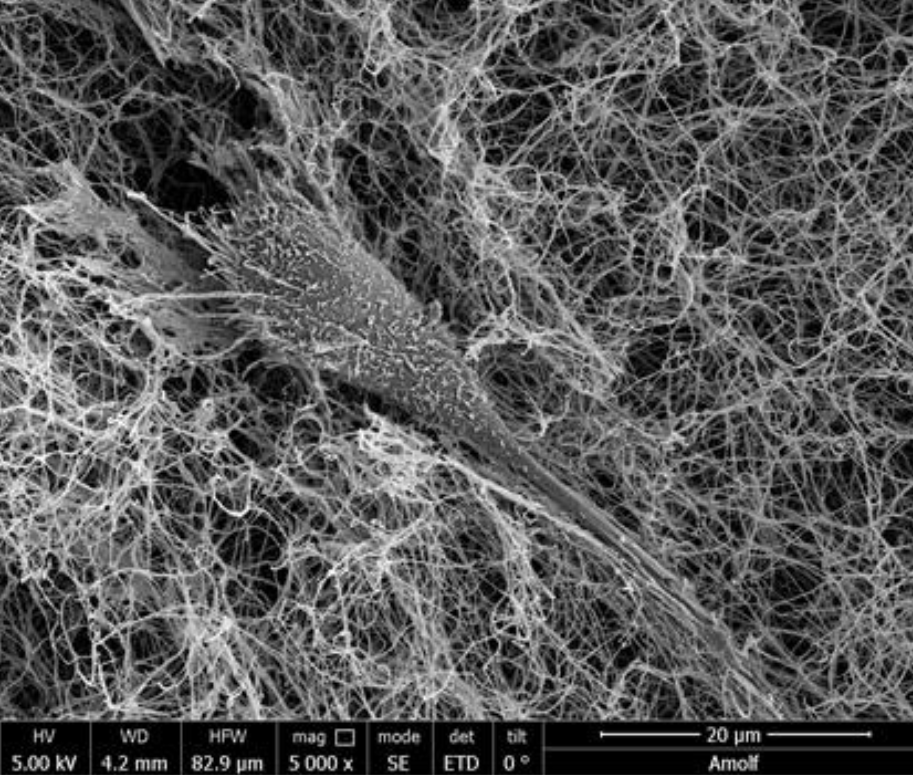
Cells actively sense and process mechanical information that is provided by their extracellular environment to make decisions on growth, motility, and differentiation. Matrix mechanics can therefore be exploited to program stem cell differentiation for organ-on-chip and regenerative medicine applications. Using a combination of mechanical measurements (rheology, optical tweezer force spectroscopy, AFM indentation) and imaging, our lab studies the bi-directional mechanical interplay between cells and reconstituted extracellular matrices that mimic different tissue environments such as liver or cartilage.
Contact: Iain Muntz
Collaborator: Prof. Gerjo van Osch (Connective Tissue Repair lab, ErasmusMC, Rotterdam)
Representative publications:
- M.T.J.J.M. Punter, B.E. Vos, B.M. Mulder and G.H. Koenderink, Poroelasticity of (bio)polymer networks during compression: theory and experiment, Soft Matter 16, 5: 1298-1305 (2020).
- D. Visscher, A. Gleadall, J. Buskermolen, F. Burla, J. Segal, G.H. Koenderink, M. Helder, P. van Zuijlen, Design and Fabrication of a Hybrid Alginate Hydrogel/poly(ɛ-caprolactone) Mold for Auricular Cartilage Reconstruction, Journal of Biomedical Materials Research Part B: Applied Biomaterials, 107:1711-1721 (2019)
- R. Staneva, F. Burla, G. H. Koenderink, S. Descroix, D.M. Vignjevic, Y. Attieh and M. Verhulsel, A new biomimetic assay reveals the temporal role of matrix stiffening in cancer cell invasion, Mol. Biol. Cell 29, 25: 2979-2988 (2018)
- K.A. Jansen, R.G. Bacabac, I.K. Piechocka and G.H. Koenderink, Cells actively stiffen fibrin networks by generating contractile stress, Biophys. J. 105: 2240-2251 (2013)