Interfacial Bonding
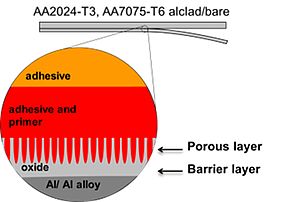
Interfacial bonding of organic coatings and adhesives on metal (oxide) surfaces
The adhesion and corrosion resistance of polymer/oxide/metal interphases in functional composite materials is an extremely important aspect of engineering in a wide variety of industries such as construction, automotive and aerospace industry. Interphases in polymer/metal joints have to withstand high mechanical forces and corrosive attack over long times to protect the functional properties of the composite. It is widely recognized that factors like alloying element enrichment and the presence of intermetallic precipitates at the metal-polymer interface have a serious influence on the corrosive de-adhesion of organic layers. Despite widespread investigations over the past decades, the adhesion between metals and polymers (organic coatings and adhesives) and the loss of adhesion in the presence of an aqueous environment are still subject to further investigations.
Adhesion and de-adhesion processes are determined by a complex co-action of:
- Molecular forces at the interface (chemical bonding, van der Waals bonding, hydrogen bonding, acid-base interactions, …)
- Supramolecular arrangement of the polymeric chains within the interphase
- The surface chemistry of the metal substrate
- The mechanical properties of the metal oxide layer, the interphase and the bulk polymer phase
- The defect density within the interphase
- The electronic properties of the metal oxide and the adjacent polymer
- The hydrolytic and oxidative resistance of the interphase
- The barrier properties of the interphase
Molecular forces at polymer/oxide/metal interfaces determine the adhesion within the interface in dry and humid environments. Disperse and polar forces, hydrogen bonding, acid-base interactions and covalent bonding contribute to the molecular adhesion forces which act only over a distance of 2-3 nanometer. The resulting forces depend on the chemistry of the oxide as well as on the chemistry and supramolecular ordering of the adsorbed polymer.
Under atmospheric conditions, that means if the polymer/metal joint is exposed to moisture and/or ion containing electrolytes, the mobility of water, hydrated ions and electrons determine the kinetics of the loss of adhesion and the kinetics of corrosive de-adhesion. The decrease in adhesion force in presence of moisture is due to the strong adsorption of water molecules on oxides and the thereby induced replacement of adsorbed polymeric chains. The incorporation of hydrated ions due to their mobility along the interface between the oxide covered metal and the polymer leads to the establishment of a sharp potential drop at the buried interface and thereby accelerates electrochemical reactions. The electron transfer from the metal through the oxide to adsorbed oxygen molecules determines the kinetic of the oxygen reduction at the interface.
Both a fundamental investigation at the molecular level using model molecules simulating the functional groups of organic coatings and a macroscopic view on adhesion and corrosive de-adhesion properties on a variety of tailored base metal/metal oxide surfaces is crucial.
The nature and type of base metallic substrate depends on the application areas, e.g. for aerospace and automotive industry, specific aluminium alloys and galvanized steels are of particular interest respectively. For the evaluation of transport phenomena theoretical models can be applied from the analysis of the physico-chemical properties of complex polymer interfaces, the application of advanced spectroscopic methods and the theoretical calculation of transport phenomena in thin films.