Active Protective Coatings
Multifunctional and eco-friendly corrosion inhibitors and evaluation of smart coatings
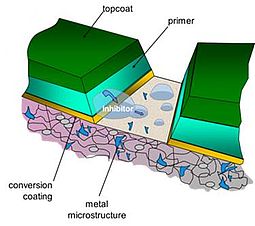
The corrosion protective properties of organic coatings can be enhanced by the use of anti-corrosion pigments in the coating. Excellent hexavalent chromium based, anti-corrosion pigments have long been available, but the health issues associated with the use of chromates have become increasingly highlighted in recent years. This, and recent very strict and international legislative restrictions have resulted in an increasing need for the development of new pigments with reduced ecological footprints.
It would appear that the corrosion performance of the underlying metal surface is improved by the autonomous surface recovery process by the underfilm precipitation of stable inhibitor compound complexes at both the cathodic and anodic sites. These inhibitors will be inactive prior to corrosion: they will be released and activated when and only when corrosion takes place in the direct surroundings of the inhibitor position, i.e. at the coating-electrolyte interface. The pre-emptive self healing process should preferably take place by the responsive release of the multifunctional corrosion inhibitors only where and when it is needed. For novel concepts to be investigated, this smart release should be triggered by corrosion controlling parameters, in this case the formation of an underfilm electrolyte: a direct trigger.
This research area is extremely complex and multidisciplinary in nature comprising expertise in the areas of physical metallurgy, organic and physical chemistry, electrochemistry and corrosion technology. It should involve an integral approach of designing the inhibitor technology concept for corroding coated surfaces, by research into:
- Development of novel eco-friendly (multifunctional) corrosion inhibitors.
- Evaluation of their intrinsic anticorrosion performance (by rapid screening and high-throughput techniques).
- Incorporation in the organic coating.
- Release mechanism from the organic coating and control of its kinetics.
- Investigation of the interaction and deposition mechanism on the corroded surface.
- Modification of delivery systems.
Release kinetics of active corrosion inhibitive pigments are crucial in an active inhibition concept. Releasing the inhibitor too quickly provides the required protection but the component will have a reduced service life. Releasing too slowly does not provide adequate protection and will result into premature failure. Furthermore, with the implementation of a combined polymer and corrosion inhibitor healing concept, the individual and combined time frames for polymer healing and the active surface healing by corrosion inhibitive pigments is even of more importance. For example, if the polymer healing is too fast compared to that of the corrosion inhibitor release kinetics, aggressive chemicals could be trapped under the newly formed coating prior to active inhibition by surface precipitation of the active corrosion inhibitive pigments and this would even accelerate the underfilm corrosion.
Ongoing EU-funded VIPCOAT project is bridging the gap between different scale experimental investigations with multiscale modeling and machine learning investigations: