Creating chaos in battery to extend lifetime
Batteries are important in accelerating the energy transition. The electric car has made its entrance thanks to the advancement of batteries, and we need batteries to store renewable solar and wind energy until the moment when we need the energy. Currently, the most widely used battery for this purpose is the lithium-ion battery: a battery that, for its weight, can store a relatively large amount of energy and has a decent lifetime when compared to other batteries. Researchers from TU Delft now present a new breakthrough in Nature Communications and Advanced Materials that could further improve the life of these batteries.
Electrolyte lifetime
A classic problem in the world of batteries is that the electrolyte slowly but surely breaks down during charging and discharging of lithium-ion batteries, which largely determines their lifespan. The breakdown cannot actually be prevented, but if you can make the electrolyte break down in a "controlled" manner, it leads to a more stable situation where it is possible to extend the battery life. To make that happen, the research group of Marnix Wagemaker (Professor of Electrochemical Energy Storage) has made an important – and more or less accidental – discovery.
Cocktail of salts and solvents
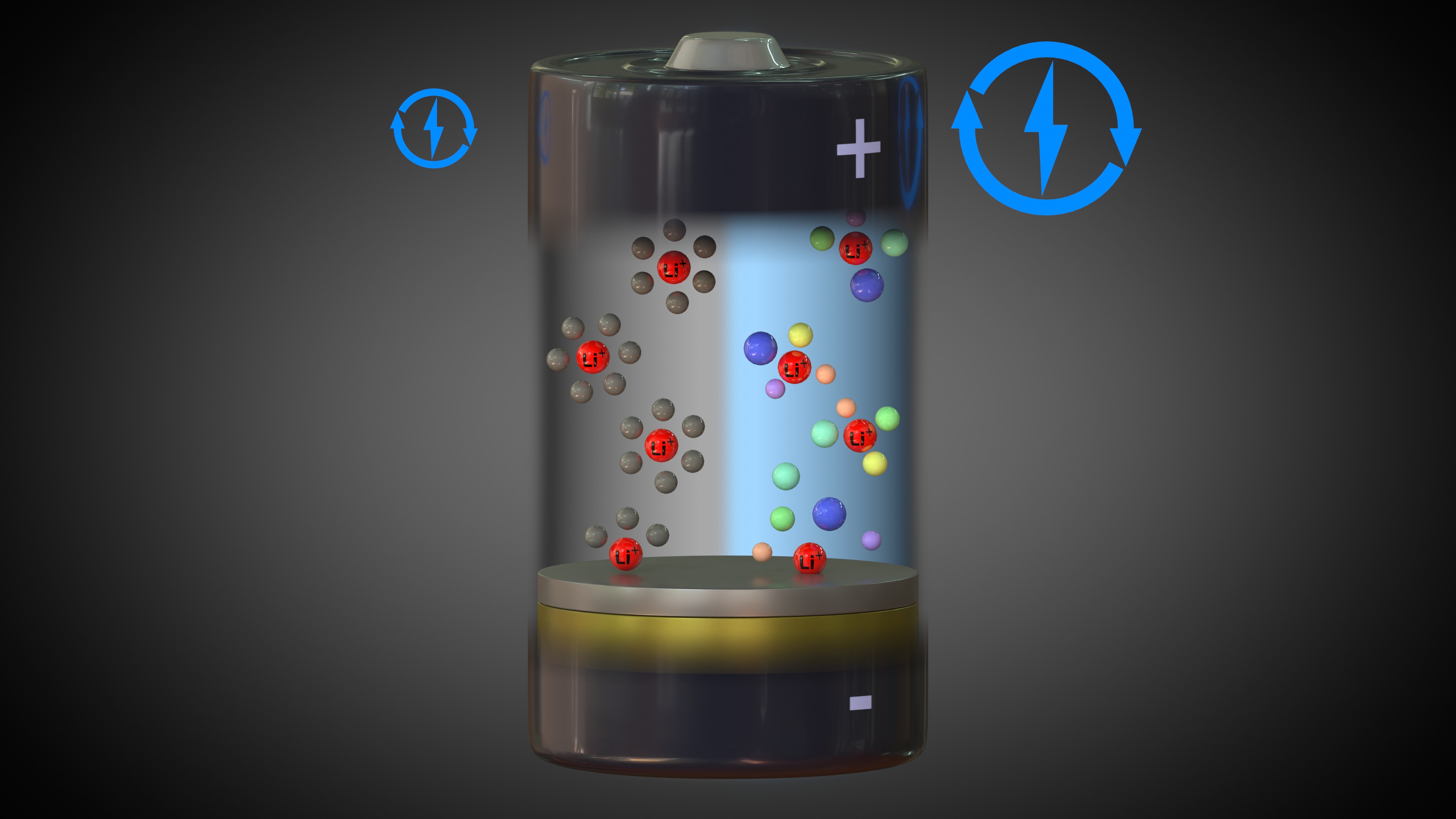
Like current lithium batteries, the new Delft battery composition consists of salts, “swimming around” in a solvent, that ensure the battery can supply power. It is a huge challenge for battery researchers to find salts and solvents that will not break down too quickly on the surface of the battery – i.e., in contact with the positive and negative electrodes – which reduces the battery's lifetime.
Using a new concept from metal alloy research, the researchers came up with the idea of mixing a cocktail of no less than five salts. This increased the chaos in the cocktail, which created a more stable surface in the battery, resulting in a substantially longer lifespan.
More information
Want to keep up to date with developments? Follow TU Delft Reactor Institute on LinkedIn. For questions, please contact our Climate & Energy Science Officer Dave Boomkens, d.j.boomkens@tudelft.nl.