A new understanding of complex carbides in creep resistant chromium steels and super alloys
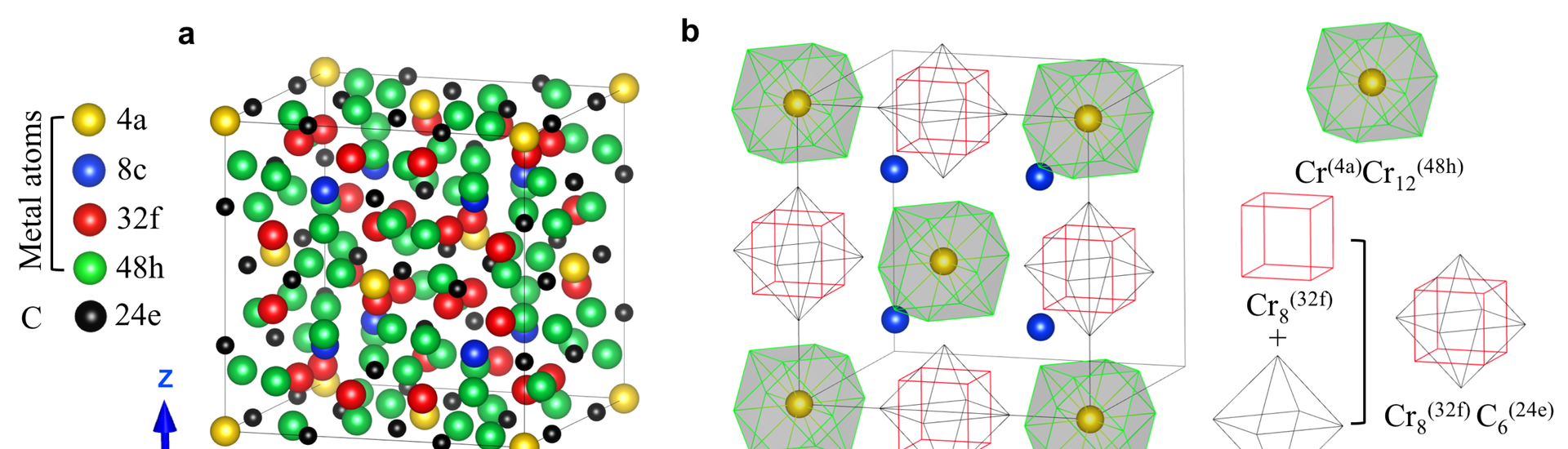
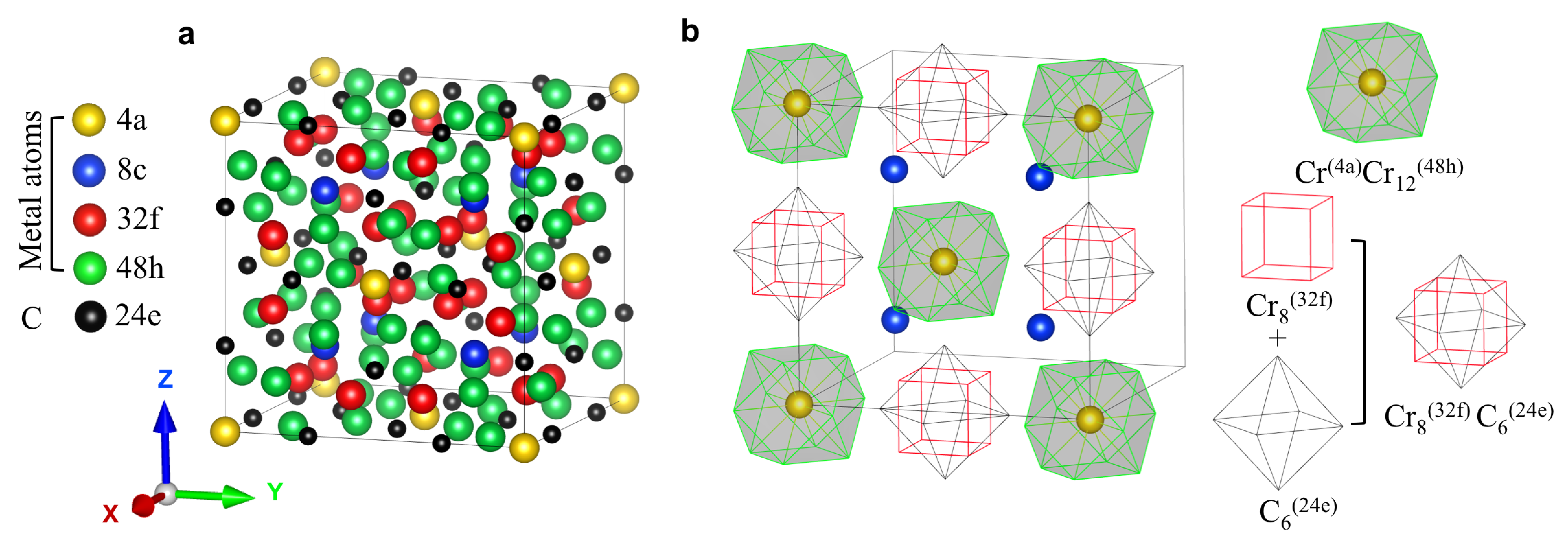
M23C6 carbides are widely used in creep resistant steels and superalloys. The thermodynamic stability of M23C6 is an important factor in retaining high strength at elevated temperatures. Although these carbides have been known for almost a century the mechanism that stabilizes the highly regular but complex structure had not been resolved. Marcel Sluiter, associate professor of computational materials science at the department of Materials Science and Engineering, together with researchers from the National Institute for Materials Science in Tsukuba, Japan, and from the Ohio State University in Columbus, USA, found that these carbides are stabilized by the formation of super-ions in a NaCl type spatial arrangement through an analysis of the electronic structure of atomic clusters within the structure. Positively charged centred rhombic dodecahedral M1M12 clusters alternate with negatively charged cuboctahedron M8C6 clusters, with additional metal atoms in the cubic interstices between these super-ions. The effect of mixed partial occupation of metal sites on the phase stability of the γ-Cr23−xFexC6 (x = 0–3) carbides is explored as function of composition and temperature. Ab initio calculations combined with statistical thermodynamics approaches reveal that the site occupation of the carbides may be incorrectly predicted when only the commonly used approach of full sublattice occupation is considered. The effect of excitations such as due to atomic vibration and Fermi level broadening also is shown to play a non-negligible role in the stability of the carbide phases. This new understanding of γ-M23C6 carbides may facilitate further development of high-chromium heat-resistant steels.
The results of the research were published earlier this week in Nature’s open access journal Scientific Reports: ‘Effect of mixed partial occupation of metal sites on the phase stability of γ-Cr23−xFexC6 (x = 0–3) carbides‘
More about <link en organisation departments materials-science-and-engineering research computational _blank>Marcel H.F. Sluiter and the computational materials science group.