‘Cohesion researchers’ unravel the mystery of hydrogen effects on materials
Hydrogen is considered a very important energy carrier with the potential to reshape the energy landscape in the future. Distributing large amounts of hydrogen requires safe steel pipelines. Steel pipelines can become brittle due to hydrogen and can therefore break. Fascinated by this urgent problem, Carey Walters (MTT), Othon Moultos (P&E) and Poulumi Dey (MSE) joined forces and turned to the cohesion programme to work on this together.
They collaborated with Abdelrahman Hussein and Gagus Ketut to investigate the cause of the brittleness, and obtain new insights into the complex underlying physical phenomena. Their results aim to improve the storage, distribution and usability of hydrogen. The results are recently published open-access in Acta Materialia and the International Journal of Hydrogen Energy.
This cohesion project is certainly laying the foundation for a more extensive and enduring partnership.
Othon Moultos
Othon Moultos (Assistant Professor Engineering Thermodynamics):
‘Poulumi, Carey and I have been working on the hydrogen storage and distribution issue for some time, albeit at different scales. Bringing together our expertise from the different fields of maritime technology, materials science and process technology seemed a logical move. As a result, we were able to investigate hydrogen and its distribution at a multi-scale level, ranging from the atomistic up to the macroscale. We gained useful knowledge about the efficient storage and resistance of high-strength steels to hydrogen embrittlement. Our research has also motivated the preparation of a new NWO proposal which is supported from important industrial stakeholders in hydrogen distribution. This cohesion project is certainly laying the foundation for a more extensive and enduring partnership.’
Abdelrahman Hussein (PostDoc Ship and Offshore Structures):
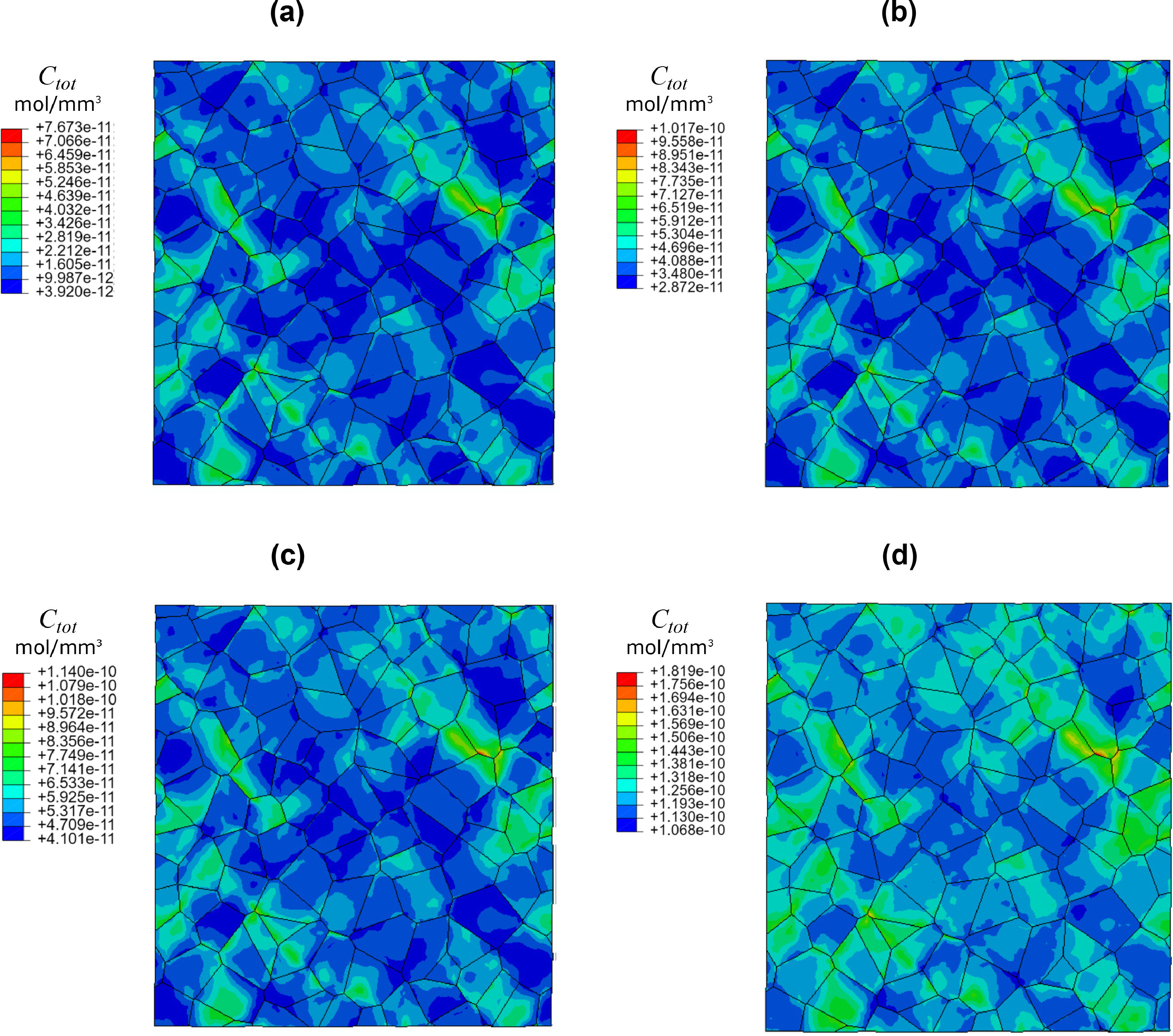
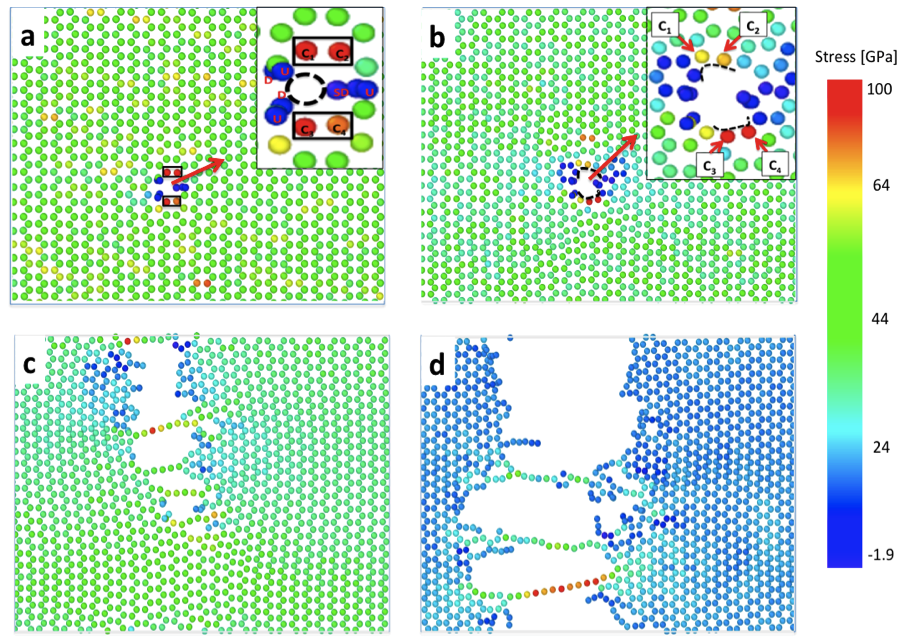
“We used RVE and crystal plasticity to show how micromechanical stresses accumulate hydrogen at grain boundaries. We also show how increasing yield strength results in higher localization of hydrogen, increasing the susceptibility to damage. This virtual framework can increase our understanding of hydrogen embrittlement and speed up developing hydrogen resistant alloys.”
Read the open-access publications:

C.L. Walters

P. (Poulumi) Dey

O. Moultos
Cohesion projects
The cohesion projects were launched in 2014 based on an idea by Dean Theun Baller, the aim of which is to encourage interdisciplinary cooperation within the faculty. A good engineer, after all, is inherently good at working with others, and most innovations take place on the cutting edge of disciplines. The cohesion projects give young researchers the opportunity to fund projects, on the condition that they work together with colleagues within the faculty and outside their own discipline. Cooperation in a large faculty like 3mE was in need of a nudge. Meanwhile, it’s paying off: Mechanical Engineering, Maritime Technology and Materials Science have proven to be extremely suitable for unexpected cross-overs with surprising outcomes.