Research
Enzyme discovery and engineering
Enzymes are the superiour catalysts of chemical reactions. The catalytic properties of novel enzymes that catalyze sugar coupling reactions and hydration reactions are studied. The multicopper oxidase laccase is studied as a model system to develop novel calorimetric techniques to measure enzyme activity on complex polymeric substrates, such as lignin.
Virtually all physical, chemical or biological processes are associated with heat effects. Quantitative measurement of the heat evolution over time can be used to monitor the progression and rate of these processes. Isothermal Titration Calorimetry has been successfully applied to measure binding of metal ions and molecules to proteins, in order to determine thermodynamic parameters and characterize different binding sites and stoichiometry. The same technique can be applied to measure enzyme kinetics, without the need to measure substrate/product levels, but instead measure the heat that is being developed (or consumed) during a chemical reaction.
Metalloproteomics in health and biotechnology
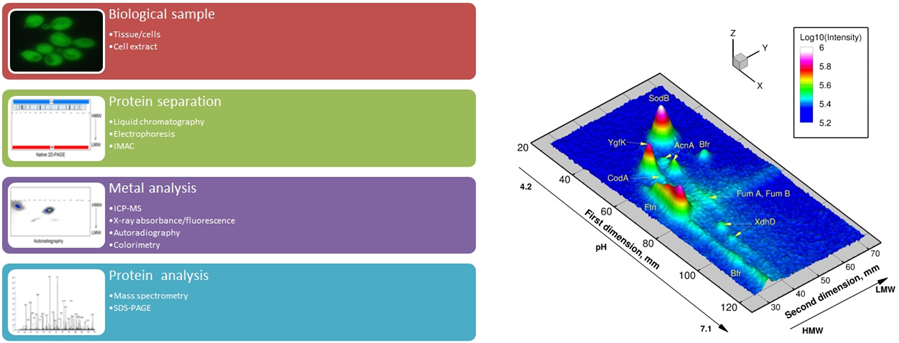
MIRAGE is a metalloproteomic technique which allows the quantitative measurement of the distribution of particular metal among different proteins in a biological samples, including the identification of the metalloproteins. MIRAGE has been used to study metal homeostasis in various microorganisms. Novel applications with relevance to metals and human health are explored.
On the left the basic components of a metalloproteomic workflow. On the right 59Fe MIRAGE of E. coli cultivated overnight at 37°C on LB medium containing 33 µM Fe.
Uncovering enzyme mechanisms
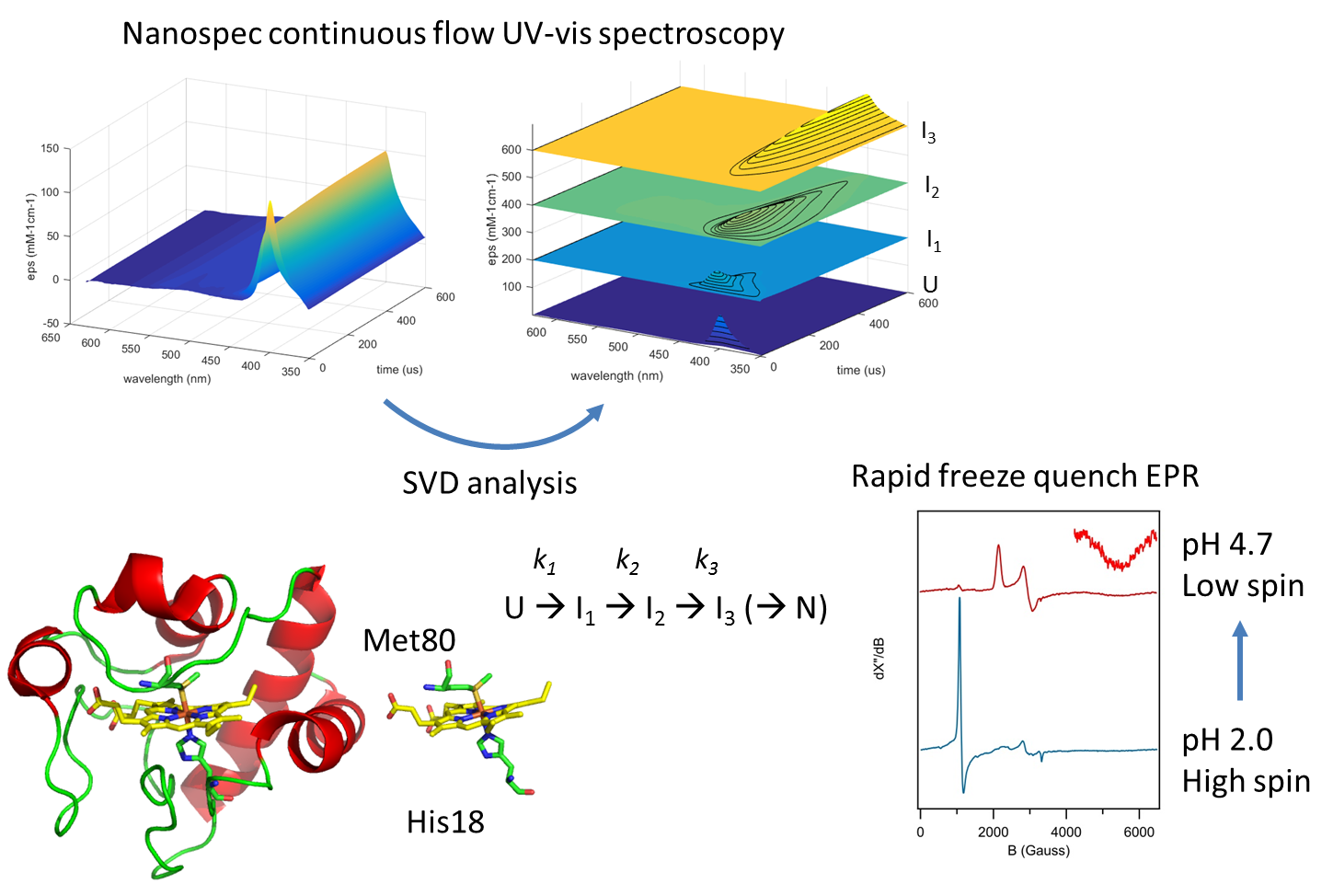
Pre-steady state, or rapid kinetics techniques, such as stopped-flow UV-vis aborbance or fluorescence spectroscopy are highly important tools to determine rate constants of individual steps in a catalytic mechanism, and to identify catalytic intermediates. The dead time of a conventional stopped flow instrument is approximately 1 ms, which does not allow the observation of intermediates with a shorter lifetime. By using a four-jet tangential micromixer we have been able to construct a continuous flow UV-vis spectroscopic device, called the Nanospec, with a dead time of 4 µs. And, using the same mixer, a freeze quench instrument called the Microsecond freeze Hyperquenching device (MHQ) with a dead time of 80 µs.